Remarkable Improvement in Plaque Psoriasis with a 4 week Short Course Liraglutide Therapy in an Obese Type 2 DM Patient
Santosh Ramakrishnan1*, Mohammed Yousuf Khan1, Anantharaman Ramakrishnan2, Shanmugasundar Gopal3, Rohit Warrier4
1Consultant Endocrinologist & Diabetologist, Magna centres for Obesity, Diabetes & Endocrinology, Hyderabad, India
2Consultant Endocrinologist & Diabetologist, Magna centres for Obesity, Diabetes & Endocrinology, Bangalore, India
3Consultant Endocrinologist & Diabetologist, Magna centres for Obesity, Diabetes & Endocrinology, Chennai, India
4Consultant Diabetologist, Magna centres for Obesity, Diabetes & Endocrinology, Bangalore, India
Abstract
Psoriasis is an immune mediated chronic skin disease associated with components of metabolic syndrome like obesity and type-2 diabetes. Previously, anti-diabetic drugs especially insulin sensitizers (metformin and pioglitazone) have shown positive outcomes in subjects with psoriasis1. Recently, many case series and longitudinal observational studies previously have demonstrated improvement in psoriasis with GLP1 agonist therapy when followed up for 8-12 weeks2,3. We report a patient with psoriasis and Type2 DM in whom a marked improvement in psoriasis was seen with liraglutide therapy, even with a short course of therapy for 4 weeks, which has not been previously recorded, to the best of our knowledge. This could be due to our subject possibly being a better GLP-1 responder based on baseline characteristics of relatively higher BMI and HbA1c4.
Case report
A 42 year old male patient with type-2 diabetes and hypertension of 5 years, presented to us for the care of his diabetes. His BMI was 32.5 kg/m2 at the time of presentation. He had a history of plaque psoriasis of 13 years duration. His PASI(Psoriasis area and severity index) was calculated as 10.2, which is Grade 2 category as per the scoring system. He was on Metformin 1 gm BD, Glimepride 2 mg BD for diabetes and Telmisartan 40 mg OD for hypertension. The patient was on topical therapy with an emollient and Mometasone and 3% salicylic acid on inflamed lesions for his psoriasis. His psoriatic lesions were stable for the previous 6 months (Fig.1). Investigations showed FPG 132 mg/dl; PPPG 228 mg/dl and HbA1c of 8.4% ; TSH was 1.32 mIU/ml. Lipids and hepato-renal parameters were normal. Liraglutide 0.6 mg subcutaneous OD was added to achieve glycemic control. The dose was up titrated to 1.2 mg OD after a week. The patient was followed up after 4 weeks. Glucose levels came down to FPG 102 mg/dl ; PPPG 142 mg/dl with a weight loss of 3 kg. Remarkable improvement was seen in severity of symptoms and the psoriatic lesion (Fig.2).The PASI score improved substantially from 10.2 to 4.7 a PASI50 response (table 1).
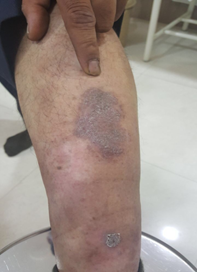
Figure 1: (before liraglutide initiation).
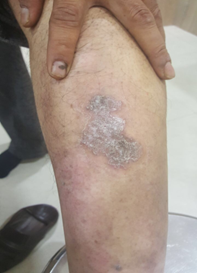
Figure 2: (after 4 weeks liraglutide therapy)
Table 1:
| Visit | FPG (mg/dl) | PPG (mg/dl) | Weight loss (kg) | PASI score (PASI50) | Liraglutide dose (mg) |
|---|---|---|---|---|---|
| Baseline | 132 | 228 | - | 10.2 | 0.6 |
| 4 weeks | 102 | 142 | 3 | 4.7 | 1.2 |
Discussion
PASI50 improvement has been shown to be a meaningful marker of clinical outcomes in chronic psoriasis, based on the quality of life and histological parameters as per various clinical trials5. Here, the difference observed over a 1 month therapy with liraglutide, in an otherwise chronic disease is attributable to drug therapy or metabolic improvement in this illustrative report. Anecdotal Case series and observational studies have previously documented this benefit, not just with liraglutide but also other insulin sensitizers, including metformin and thiazolidinediones1,6,7. Most of these studies have observed a positive clinical outcome only after 8 to 12 weeks therapy with liraglutide; in our case report, the betterment was observed on completion of 4 weeks of therapy itself. Subjects with higher baseline BMI and HbA1c values have shown to respond better to the effects of GLP-1 analogues, explaining the faster and more efficacious response seen with our case4. GLP-1 analogues as a class have shown improvement in psoriatic skin lesions; this has been speculated to be due to the drugs action on iNKT (invariant Natural Killer T cells) that have shown to play a role in disease pathogenesis8. Thus, we advocate further for the use of liraglutide therapy for psoriasis, especially for those subjects who possibly could be better responders based on baseline characteristics.
References
- Singh S, Bansali A. “Randomized placebo control study of insulin sensitizers (Metformin and Pioglitazone) in psoriasis patients with metabolic syndrome (Topical Treatment Cohort),”. BMC Dermatology 16. 2016; article number 12.
- Faurschou A, Gyldenløve M, Rohde U, et al.“Lack of effect of the glucagon-like peptide-1 receptor agonist liraglutide on psoriasis in glucose-tolerant patients--a randomized placebo-controlled trial,”. J Eur Acad Dermatol Venereol. 2015; 29: 555–559.
- Carlin CS, Feldman SR, Krueger JG, et al. “A 50% reduction in the Psoriasis Area and Severity Index (PASI 50) is a clinically significant endpoint in the assessment of psoriasis,”. J Am Acad Dermatol. 2004; 50: 859–866.
- Babenko AY, Savitskaya DA, Kononova YA, et al. “Predictors of Effectiveness of Glucagon-Like Peptide-1 Receptor Agonist Therapy in Patients with Type 2 Diabetes and Obesity,”. Journal of Diabetes Research. 2019.
- Xu X, Lin L, Che P, et al. “Treatment with liraglutide, a glucagon-like peptide-1 analogue, improves effectively the skin lesions of psoriasis patients with type 2 diabetes: A prospective cohort study,”. Diabetes Research and Clinical Practice. 2019; 150: 167–173.
- Faurschou A, Knop F, Thyssen J, et al. Improvement in psoriasis after treatment with the glucagon-like peptide-1 receptor agonist liraglutide. Acta Diabetol. 2014; 51: 147–150.
- Ahern T, Tobin A, Corrigan M, et al. Glucagon-like peptide-1 analogue therapy for psoriasis patients with obesity and type 2 diabetes: A prospective cohort study. J Eur Acad Dermatol Venereol. 2013; 27: 1440–1443.
- Hogan AE, Tobin AM, Ahern T, et al. “Glucagon-like peptide-1 (GLP-1) and the regulation of human invariant natural killer T cells: lessons from obesity, diabetes and psoriasis,”. Diabetologia volume. 2011; 54: Article number: 2745.
