Hydroxychloroquine (HCQS) for glycaemic control: a remarkable turnaround in a patient with Type 2 DM
Santosh Ramakrishnan1*, Anantharaman Ramakrishnan2, Rohit Warrier3, Shanmugasundar Gopal4
1Consultant Endocrinologist & Diabetologist, Magna centres Clinics for Obesity, Diabetes & Endocrinology, Hyderabad, India
2Consultant Endocrinologist & Diabetologist, Magna centres Clinics for Obesity, Diabetes & Endocrinology, Bangalore, India
3Consultant Diabetologist, Magna centres Clinics for Obesity, Diabetes & Endocrinology, Bangalore, India
4Consultant Endocrinologist & Diabetologist, Magna centres Clinics for Obesity, Diabetes & Endocrinology, Chennai, India
Abstract
We describe a case where we effectively managed chronic, uncontrolled type 2 diabetes with hydroxychloroquine (HCQS) as an add on therapy, wherein the patient was on multiple oral hypoglycemic agents along with a combination of injectable incretin/insulin therapy prior to HCQS initiation. Six months into combination therapy with HCQS, the target HbA1c was achieved for the first time in the patient’s recent history at a much lower daily insulin dose (56% requirement drop), which has never been documented before. This combination also resulted in significant weight loss. We make a case for advocating the use of HCQS to the available routine diabetes therapeutic agents, especially if the blood sugars fail to achieve target levels in spite of being on intensive management with insulin and for the obese phenotype.
Introduction
India reports the second highest prevalence of diabetes after China among Southeast Asia region, with a staggering number close to 70 million (Bajaj, S, 2018). Considering the higher risk for the Indian population, with the higher baseline leptin levels, higher insulin levels, “thin-fat Indian” or “sarcopenic obesity” it becomes essential to have drugs that are cost efficient. This is probably the role HCQS could play in managing diabetes in the Asian-Indian context. From 2014, HCQS has been approved by DCGI, as an add on agent with diet and exercise, to patients who are inadequately controlled with metformin and sulfonylurea combination1.
Case report
A 48 year old gentleman presented to our centre with uncontrolled type 2 diabetes for 12 years at the time of presentation. At his baseline visit his BMI was 30 kg/m2 and presented with an HbA1c of 9.1%. Subject was infrequent with his daily exercise although he was more careful with his diet. This was the probable reason he had chronic weight gain issues, especially being on insulin therapy. He was on intensive therapy consisting of twice daily premix insulin (30/70 biphasic aspart) of 15 units, along with 6 mg of glimepiride, 2 g of metformin and 50 mg of sitagliptin daily. Considering his need for achieving better glycemic control, his daily insulin dose was gradually stepped up to (1 U/kg body weight as basal bolus therapy). In spite of this, his diabetes remained uncontrolled. At his second office visit, he was started on 25 mg of empagliflozin. Six months post initiation of SGLT-2 inhibitors, patient presented with minimal weight loss (0.5 kg) and HbA1c of 9%.
Considering his minimal change in glycemic parameters and insignificant weight loss, he was switched from sitagliptin to GLP-1 analogue dulaglutide 1.5 mg once weekly, along with rest of the oral hypoglycemic agents. We did not consider adding thiazolidinediones to his current regimen considering the possibility of further weight gain and the considerable risk of edema and heart failure with combined insulin and glitazone therapy2. He was advised to closely monitor and follow up with us frequently. Substantial effort was also made to correct lifestyle factors with exercise and diet.
He presented to our centre a year later with a HbA1c of 7.9% with the on-going prescribed therapy, this was deemed unsatisfactory, given the substantial cost of therapy. We initiated hydroxychloroquine as an add on therapy, considering the recent prospective and retrospective data showing the beneficial effects of the same in managing dysglycemia3. Hydroxychloroquine (HCQ) has been approved as add on therapy for patients with Type2 DM India since 2015, for subjects who fail to achieve glycemic target in spite of being on multi drug therapy4. Subsequently into therapy 3 months post initiation of HCQ 400 mg/daily along with insulin and rest of the oral hypoglycemic agents, he achieved a target HbA1c of 6.8% (table 1). Much to ours and the satisfaction of patient; never had the patient reported to have achieved this value over last 7 years. Further 3 months into therapy, patient maintained an average HbA1c of 7% along with an additional weight loss of 3 kg, at a much lower total daily dose of insulin than prior to initiating hydroxychloroquine (Fig.1).
Table 1:
| Visit | Months | Oral hypoglycemic agent (daily dose) | Insulin dose (units/day) | HbA1c (%) | HbA1c (mmo/l) | Weight (kg) |
|---|---|---|---|---|---|---|
| 1 | 0 | Glimepiride (6 mg)/Metformin (2 g)/Sitagliptin (50 mg) | 30 | 9.1 | 11.9 | 100 |
| 2 | 3 | Glimepiride (6 mg)/Metformin (2 g)/Sitagliptin (50 mg)/Empagliflozin (25 mg) | 60 | - | - | 100.5 |
| 3 | 9 | Glimepiride (6 mg)/Metformin (2 g)/Dulaglutide (1.5 mg/weekly)/Empagliflozin (25 mg) | 60 | 9.0 | 11.7 | 100 |
| 4 | 21 | Glimepiride (6 mg)/Metformin (2 g)/Dulaglutide (1.5 mg/weekly)/Empagliflozin (25 mg)/HCQ (400 mg) | 54 | 7.9 | 10 | 98 |
| 5 | 24 | Glimepiride (6 mg)/Metformin (2 g)/Dulaglutide (1.5 mg/weekly)/Empagliflozin (25 mg)/HCQ (400 mg) | 30 | 6.8 | 8.2 | 96 |
| 6 | 27 | Glimepiride (6 mg)/Metformin (2 g)/Dulaglutide (1.5 mg/weekly)/Empagliflozin (25 mg)/HCQ (400 mg) | 24 | 7.0 | 8.6 | 93 |
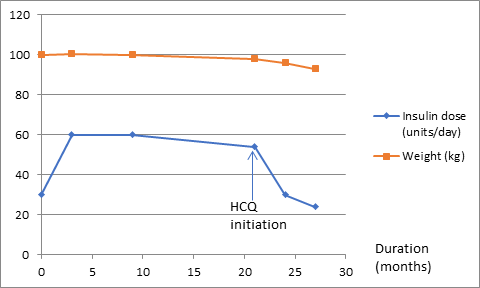
Figure 1: (insulin requirement and weight changes)
Conclusion
Now the evidence is clearly showing that diabetes is an inflammatory disease, and that’s where the role of anti-inflammatory agents could have importance in the management of diabetes. HCQ is a long tested agent that has been tested and proven to be very efficacious in diseases of inflammatory origin, like Lupus and RA, thereby playing a possible role in management of diabetes. Recent reports have suggested that in subjects with higher baseline hsCRP levels, who are initiated with HCQS5, the reduction in HBA1c is higher compared to those who are otherwise. This could be the best explanation in our case, since the subject was an obese gentleman, which itself is a pro-inflammatory condition, hence the significant response when HCQS was complemented with SGLT-2 inhibitors/GLP-1 analogues, resulting in robust weight loss and lower insulin requirement. Prior few trials have also shown this benefit in terms of HbA1c reduction primarily6.
This suggests the possibility of considering HCQS as part of our available diabetes armamentarium, especially considering its action as a blood glucose lowering and an anti-inflammatory agent, both of which are pathophysiological processes in diabetes. This drug is generally well tolerated with few mild gastrointestinal side effects including nausea, and vomiting. Maculopathy and retinopathy have been rarely reported with this drug; this has been observed if the dosage is much higher than recommended. This drug could especially be useful in “sarcopenic obese,” high dose insulin requiring Asian phenotypes, where the underlying inflammatory disease process plays a larger pathophysiological role than their western counterparts.
References
- Bajaj S. “RSSDI clinical practice recommendations for the management of type 2 diabetes mellitus 2017”. Int J Diabetes Dev Ctries. 2018 Mar; 38(Suppl 1): 1–115.
- Singh S, Loke YK, Furberg CD. “Thiazolidinediones and heart failure a teleo-analysis”. Diabetes Care. 2007; 30: 2148-53.
- Hage M, Al-Badri M, Azar S. “A favorable effect of hydroxychloroquine on glucose and lipid metabolism beyond its anti-inflammatory role”. Ther Adv Endocrinol Metab. 2014 Aug; 5(4): 77–85.
- Luthra A, Misra A. “The marketing of unproven drugs for diabetes and dyslipidaemia in India”. The Lancet Diabetes & Endocrinology. 2015; 3.
- Pareek A. “AACE 2018 Type 2 Diabetes Patients Respond to Hydroxychloroquine”. AACE. 2018.
- Pareek A, Chandurkar N, Thomas N, et al. “Efficacy and safety of hydroxychloroquine in the treatment of type 2 diabetes mellitus a double blind randomized comparison with pioglitazone”. Current Medical Research and Opinion. 2014; 30(7).
